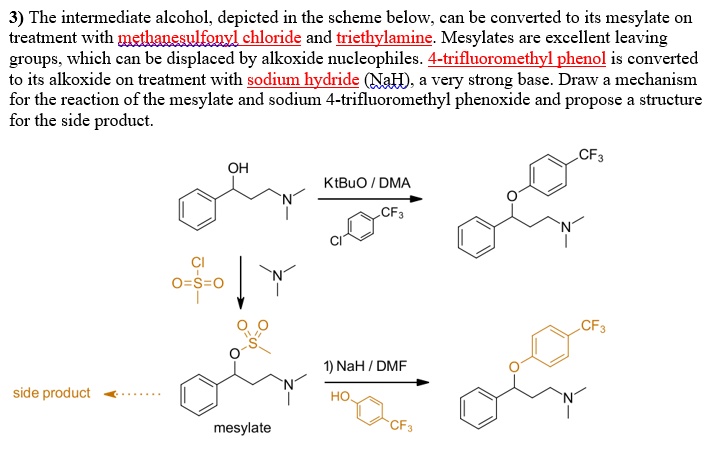
Triethylamine Organic Base Molecule Skeletal Formula Stock Vector (Royalty Free) 1128981863 | Shutterstock
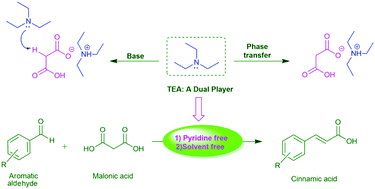
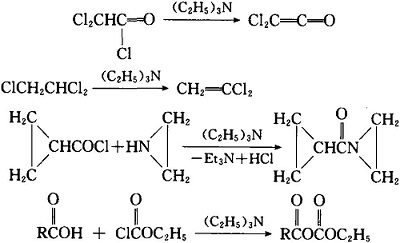
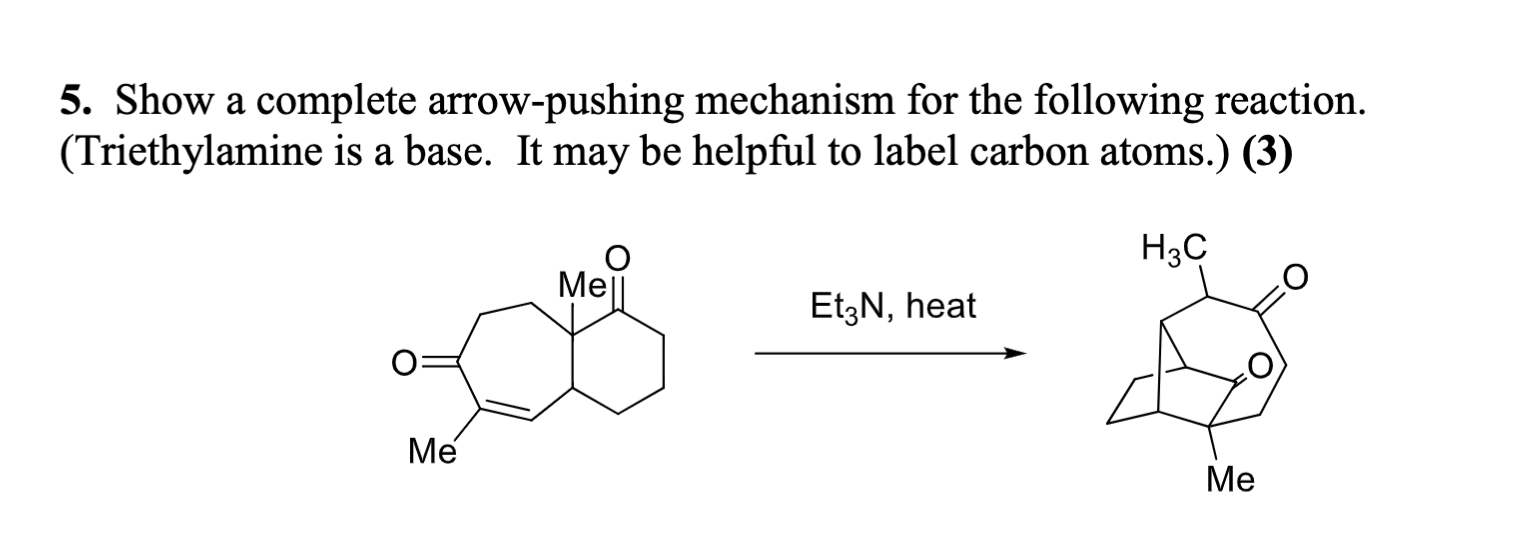
Triethylamine: a potential N-base surrogate for pyridine in Knoevenagel condensation of aromatic aldehydes and malonic acid - New Journal of Chemistry (RSC Publishing)
Triethylamine organic base molecule. 3D rendering. Atoms are represented as spheres with conventional color coding: hydrogen (white), carbon (grey), n Stock Photo - Alamy
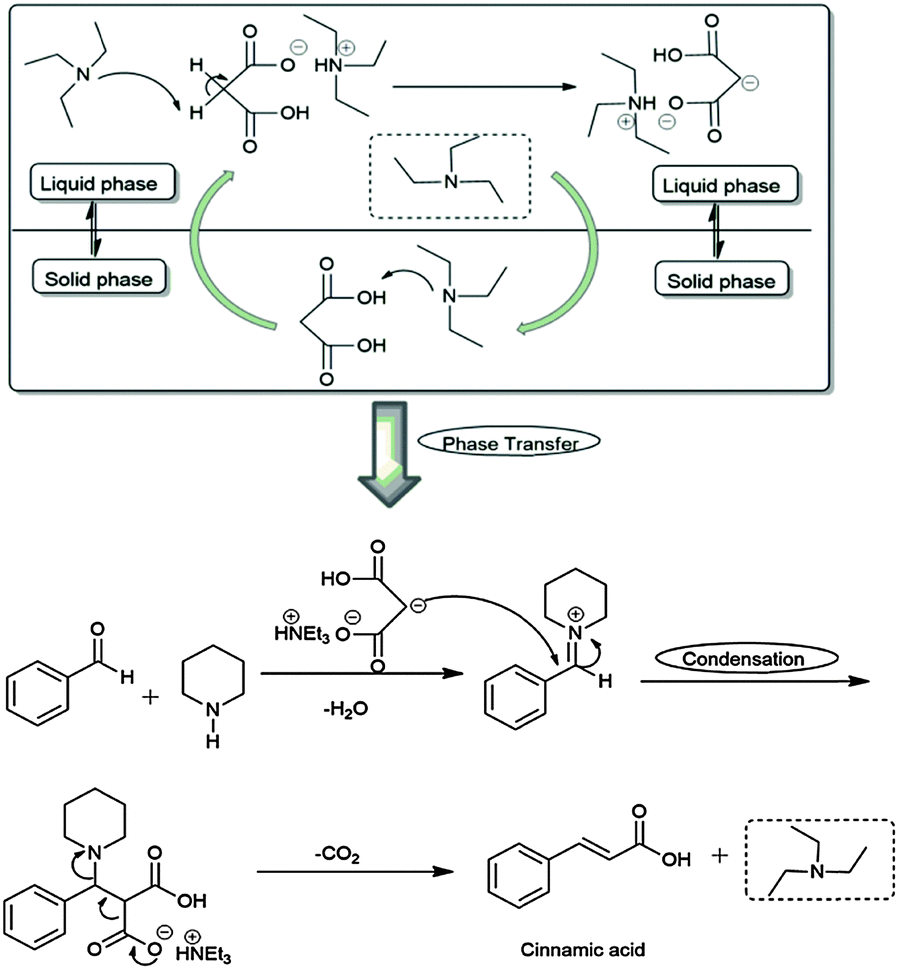
Triethylamine: a potential N-base surrogate for pyridine in Knoevenagel condensation of aromatic aldehydes and malonic acid - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C5NJ03125G